Index ntawm nplooj ntawv ntsiab lus
En Ok Pauv Reform Peb xav qhia rau koj hauv qab no ncej, uas cuam tshuam nrog Qhov pH ntawm lub pas dej da dej yog dab tsi thiab yuav tswj tau li cas
Dab tsi yog pH, qhov tseem ceeb, qhov tseem ceeb thiab ntsuas nws li cas

Dab tsi yog pH
pH yog ib qho kev ntsuas ntawm acidity los yog alkalinity ntawm cov kua aqueous.
Yog li ntawd, raws li peb tau hais, PH yog ib qho kev ntsuas ntawm acidity los yog alkalinity ntawm sutantica uas qhia txog cov concentration ntawm hydrogen ions tam sim no nyob rau hauv tej yam kev daws teeb meem.
Lub ntsiab lus pH sawv cev rau dab tsi?
- Ntawm qhov tod tes, hais qhov ntawd Lub ntsiab lus pH sawv cev rau lub peev xwm ntawm hydrogen lossis peev xwm ntawm hydrogen ions.
PH yog dab tsi | Basic Chemistry
Piav video hais txog tus nqi ph txhais li cas
Qhov sib npaug ntawm pH qhov tseem ceeb hauv tib neeg lub cev
Pas dej pH theem

Dab tsi yog pH ntawm cov dej hauv pas dej
pas dej pH txhais li cas?

Qhov pH ntawm lub pas dej txhais li cas?
Lub pH ntawm lub pas dej ua ke yog
Lub pH ntawm lub pas dej yog dab tsi: Lub pH yog lub peev xwm ntawm hydrogen, tus nqi uas sib haum mus rau qhov concentration ntawm hydrogen ions hauv dej ntawm koj lub pas dej ua ke thiab yog li ntawd yog cov coefficient uas qhia txog qib acidity los yog qhov tseem ceeb ntawm cov dej. . Yog li, pH yog tus saib xyuas qhia qhov concentration ntawm H + ions hauv dej, txiav txim siab nws cov kua qaub lossis cov cim tseem ceeb.
Qhov zoo tshaj plaws pas dej pH tus nqi
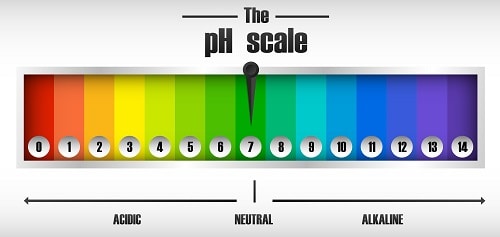
Lub pH nplai mus los ntawm 1 txog 14, nrog pH 7 yog ib qho kev daws teeb meem nruab nrab.
Tus pH yog tus nqi uas qhia nrog lub logarithmic nplai ntawm qhov muaj nuj nqis 0 thiab 14.
Yog li ntawd, txhawm rau ntsuas lub acidity ntawm cov kua, thiab nyob rau hauv peb cov ntaub ntawv ntawm cov dej pas dej ua ke, tshuaj thiab tam sim no peb yuav siv. pH nplai uas suav nrog cov nqi ntawm 0 txog 14.
pas dej zoo pH
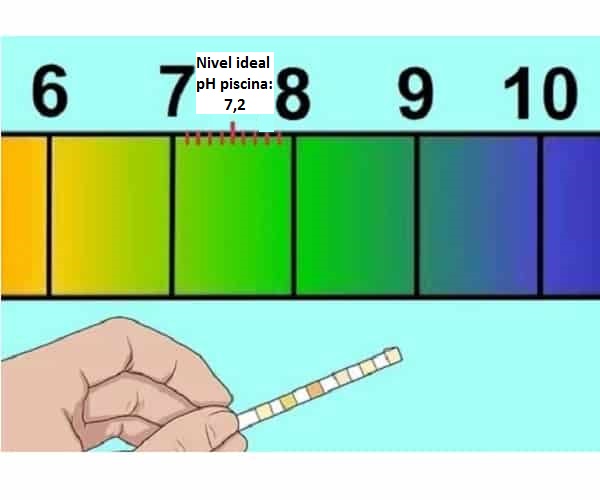
Pas dej pH: yog ib qho tseem ceeb tshaj plaws hauv kev tu pas dej.
Tus nqi tsim nyog rau lub pas dej da dej pH: nruab nrab ntawm 7.2 thiab 7.6 qhov zoo tagnrho ntawm nruab nrab pH.

Yog li ntawd, muaj pH nyob rau hauv qhov ntau no tsis yog tsuas yog zoo rau muaj dej nyob rau hauv qhov pom kev zoos txij thaum pH qis los yog siab considerably txo cov tshuaj tua kab mob, tab sis nws kuj yog zoo tagnrho rau cov tawv nqaij thiab qhov muag ntawm da dej.
pas dej saline pH

ph saline pas
- Tiag tiag, cov ph laug los ua tib yam li cov pas dej uas tau kho nrog cov tshuaj chlorine txij li kev saib xyuas ntawm cov pas dej siv ntsev kuj yuav tsum tau saib xyuas tsis tu ncua ntawm cov pH ntawm dej.
- Yog li ntawd, pH ntawm ntsev pas dej yuav tsum muaj ib tug pH nyob nruab nrab ntawm 7 thiab 7,6, qhov zoo tagnrho yog nyob nruab nrab ntawm 7,2 thiab 7,4.
Vim li cas pH ntawm cov dej hauv pas dej tseem ceeb heev?
Acid, nruab nrab thiab alkaline pH qhov tseem ceeb
Kev faib tawm ntawm qhov ntsuas pH tus nqi
Dab tsi yog cov nqi pH

Lub pH nplai mus los ntawm 1 txog 14, nrog pH 7 yog ib qho kev daws teeb meem nruab nrab.
Yog li, nws hloov tawm tias pH yog tus nqi uas tau hais tawm ntawm qhov ntsuas logarithmic nruab nrab ntawm qhov tseem ceeb 0 (tsis tshua muaj acidic) thiab 14 (alkaline heev); Hauv nruab nrab peb pom tus nqi 7 cataloged li nruab nrab.
pH scale universal pH qhia
Nws txhais li cas tias cov khoom muaj acidic lossis alkaline pH?
Dab tsi yog acids thiab bases?
Cov kua qaub thiab cov hauv paus yog cov khoom uas muaj nyob hauv qhov xwm txheej thiab txawv ntawm lawv qib pH, uas yog, los ntawm lawv qib acidity lossis alkalinity. Kev txiav txim siab ntawm seb cov khoom puas yog acidic lossis alkaline yog tswj hwm los ntawm qib acidity lossis alkalinity ntsuas los ntawm pH nplai thiab thaj tsam ntawm 0 ( acidic heev mus rau 14 (tsis tshua muaj alkaline). txawm li cas los xij muaj ntau yam kev lag luam thiab tib neeg siv.
Cov tshuaj acidic yog dab tsi?
- acid pH: pH tsawg dua 7
Nws txhais li cas tias tus nqi pH yog acidic?
- Hais tias ib yam khoom yog acidic txhais tau hais tias nws yog nplua nuj nyob rau hauv H+ (hydrogen ions): pH ntau dua 7
- Vim li no, Acids yog cov khoom uas muaj pH tsawg dua 7. (pH ntawm dej sib npaug li 7, suav tias yog nruab nrab), uas nws chemistry feem ntau muaj cov hydrogen ions ntau thaum ntxiv dej. Lawv feem ntau hnov mob nrog lwm yam tshuaj los ntawm kev poob protons (H+).
Dab tsi yog cov khoom nruab nrab?
- Nruab nrab pH tus nqi: pH sib npaug rau 7-
Nws txhais li cas tias pH tus nqi nruab nrab?
- pH yog ib qho kev ntsuas ntawm acidic/basic dej.
- Qhov ntau yog los ntawm 0 txog 14, nrog 7 yog nruab nrab.
Cov tshuaj alkaline yog dab tsi?
- Cov khoom uas muaj lub hauv paus lossis alkaline pH: pH ntau dua 7.
Nws txhais li cas thaum tus nqi pH yog alkaline?
- Tias ib yam khoom yog alkaline txhais tau hais tias nws tsis zoo hauv H+ (los yog nplua nuj hauv OH hauv paus-, uas neutralize H+).
- Rau tag nrho cov no, Bases, ntawm qhov tod tes, yog cov khoom uas muaj pH ntau dua 7., uas nyob rau hauv aqueous daws feem ntau muab hydroxyl ions (OH-) nyob nruab nrab. Lawv zoo li muaj zog oxidants, uas yog, lawv hnov mob nrog protons los ntawm ib puag ncig nruab nrab.
Qhov sib txawv ntawm pH thiab pOH tus nqi
Qhov sib txawv ntawm tus nqi ntawm pH thiab pOH yog dab tsi
Tus nqi pH li cas yog dab tsi?
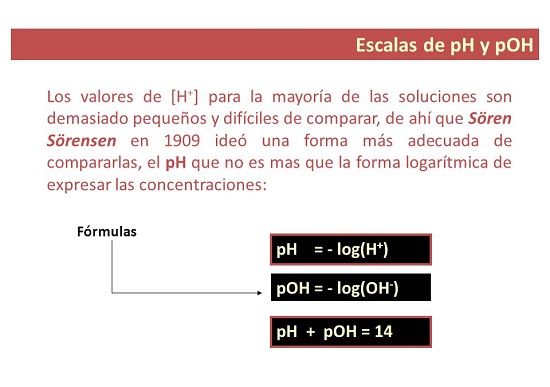
- Hauv ib txoj kev, pH yog qhov ntsuas qhov ntawd siv los tsim cov qib ntawm acidity lossis alkalinity ntawm kev daws. Lub "p" sawv cev rau "muaj peev xwm", uas yog vim li cas pH hu ua: muaj peev xwm ntawm hydrogen.
Tus nqi pOH yog dab tsi?
- Rau koj ib feem. pOH yog ib qho kev ntsuas ntawm qhov concentration ntawm hydroxyl ions hauv cov tshuaj. Nws yog qhia raws li lub hauv paus 10 tsis zoo logarithm ntawm hydroxyl ion concentration thiab, tsis zoo li pH, yog siv los ntsuas cov alkalinity theem ntawm kev daws.
Dab tsi yog qhov tseem ceeb ntawm pH nplai hauv cov khoom tu?
Yuav ntsuas pH tus nqi thiab hom ntsuas li cas
Yuav ntsuas pH pas dej li cas

Ua qhov ntsuas pH hauv tsev nrog lub pob liab liab

Yuav ntsuas pH pas dej li cas, ntau npaum li cas thiab hom meters
Yuav ua li cas xam pH

Lub pH nplai yog xam los ntawm qhov tsis zoo logarithm.
Tus nqi pH yog logarithmic
pH yog lub logarithm Qhov concentration ntawm H ions+, nrog lub cim hloov pauv: Ib yam li ntawd, pOH txhais tau tias yog logarithm Qhov concentration ntawm OH ions-, nrog rau kev hloov pauv: Cov kev sib raug zoo hauv qab no tuaj yeem tsim los ntawm cov pH thiab pH. Pib los ntawm kev qhia ntawm cov khoom ionic ntawm dej (Kw):
Logarithmic pH Formula
- Log pH Formula: xam pH siv pH sib npaug: pH = -log[H3O+].
Nws txhais li cas tias tus nqi pH yog logarithmic
Qhov tseeb tias pH yog logarithmic txhais tau hais tias ntawm txhua chav tsev ntawm qhov ntsuas muaj qhov sib txawv ntawm 10 qhov sib txawv,
- Yog li, qhov no txhais tau hais tias pH 5 yog 10 npaug ntau acidic dua pH 6, thiab pH 4 yog 100 npaug acidic dua pH 6.
Yuav ua li cas xam pH nrog logarithms?
qhov ntsuas ntawm pH yog xam txog a logarithm tsis zoo. A logarithm qhov tsis zoo tsuas qhia tias pes tsawg zaus tus lej yuav tsum muab faib. Qhov sib npaug ntawm pH tuaj yeem sau tau raws li hauv qab no: pH = -log[H3O+]. Qee lub sij hawm qhov sib npaug yog sau li no: pH = -log[H+].
Yog vim li cas rau txoj kev loj hlob ntawm tus nqi pH: qhov pH nplai tau tsim, siv dej raws li tus qauv.

- Nws yog ib qho kev sim qhov tseeb tias tsuas yog 1 mole hauv 5,50,000,000 moles ntawm dej ionizes rau hauv ib H + thiab ib OH-.
- Qhov no yog tib qhov piv txwv li ib gram ntawm hydrogen ions hauv 10.000.000 liv dej.
- Yog li ntawd, ib liter dej muaj 1/10.000.000 (los yog) 1/107 ntawm ib gram ntawm H +. Rau kev siv txhua hnub, tsuas yog siv 'Potency' daim duab, nrog rau pH cim tso ua ntej nws.
pas dej ua ke pH tshuab xam zauv
Vim li cas pH hauv pas dej ua ke poob lossis nce?
Yuav ua li cas nce pH ntawm lub pas dej thiab yuav ua li cas yog tias qib qis
pH pas dej ua ke thiab siab pH ua rau
Yuav ua li cas yog tias pH qib siab tshaj tus nqi pom zoo?
Cov txiaj ntsig ntawm lub pas dej pH siab: Yuav ua li cas yog tias pH ntawm pas dej ua ke siab

- Ua ntej tshaj plaws, qhov pH siab ntawm lub pas dej da dej ua rau nws nyuaj rau cov dej ntws mus kom zoo thiab ntau zaus, nws yog ib qho teeb meem uas tshwm sim vim yog siv qee hom lim dej los yog lub tshuab rhaub dej.
- Cov tsos mob ntawm peb lub cev yog cov tawv nqaij qhuav thiab khaus khaus.
- Ib yam li ntawd, cov dej pos huab hloov pH ntawm lub pas dej, qee zaum los ntawm kev siv cov tshuaj chlorine tsis txaus lossis cov khoom siv niaj hnub los tua cov dej.
- Raws li yog tias tsis txaus, pH siab yuav txhawb kom tsim cov txiv qaub tso rau hauv lub pas dej uas yuav xaus nrog cov dej ntshiab. Cov kua qaub no yuav nkag mus rau hauv cov kav dej thiab lwm yam kev teeb tsa, cuam tshuam rau lawv txoj kev ruaj ntseg thiab kev ua haujlwm zoo. Lawv tseem yuav lo rau cov phab ntsa thiab cov plag tsev, hloov cov tsos thiab kev huv ntawm lub pas dej.
Hauv qab no, yog tias nws txaus siab rau koj, peb muab koj qhov txuas mus rau qhov nplooj ntawv uas peb txheeb xyuas tag nrho cov txiaj ntsig ntawm pH siab hauv cov pas dej da dej thiab lawv qhov ua tau.